Chemical-Physical Methods of Wastewater Treatment
Chemical-physical processes for wastewater treatment are used successfully in numerous industries worldwide. They are highly robust, efficient and cost-effective. Thanks to our extensive project experience, we can develop the optimum solution for you - taking into account all ecological and economic requirements.
Use of Modern Chemical-Physical Processes to Achieve a Higher Level of Pollutant-Free Status
Chemical-physical processes for wastewater treatment play a key role in protecting water resources. They are highly efficient, reliable and cost-effective and are particularly suitable for industrial wastewater containing complex substances.
As experts in industrial water treatment, we develop customized solutions for customers in the semiconductor, solar, battery, food, beverage and pharmaceutical industries, among others. These industries place high demands on wastewater quality, as process wastewater often contains a large number of dissolved and undissolved pollutants.
Our wide-ranging process portfolio is designed to solve these challenges safely and sustainably. As the composition of many wastewaters is very complex, effective purification often requires multi-stage process chains in which several chemical-physical technologies are combined. These include precipitation and flocculation, filtration, ion exchange, membrane technology and adsorption.
By intelligently combining these processes, we develop highly efficient and economical solutions that are individually tailored to the respective industries and site conditions. In this way, a wide variety of wastewater types can be reliably treated and legal requirements can be permanently complied with.
The Advantages of Using Chemical-Physical Treatment Methods at a Glance:
Efficient pollutant removalHigh efficiency in the removal of heavy metals, silicon, aluminum, fluorides, arsenic and suspended solids (particles).
FlexibilitySystem configuration & modular design of the system adapted to your needs. Design for batch process or continuous process possible.
QualityInnovative solutions that meet the highest environmental technology standards and quality requirements make us your reliable partner.
SustainabilityReduction of environmental impact and conservation of resources through water recycling, resource recovery and heat recovery.
Cost efficiencyOptimized processes for lower chemical consumption and lower investment and operating costs.
Our holistic approach includes all project phases from planning, procurement and construction to construction supervision and commissioning. We work closely with the customer to develop customized solutions.

Wastewater Treatment with Membrane Technology
With membrane filtration, dissolved and undissolved substances can be separated from the wastewater and concentrated. Separation takes place under pressure - the membrane retains particles or molecules of a certain size with a certain pore size. The various processes are used for water treatment, wastewater treatment, process water recycling and for concentrating valuable substances for recovery.
The Microfiltration is used to separate particles, bacteria and yeasts. It is therefore used for cold sterilization and the separation of oil-water emulsions, among other things.
The Nanofiltration can retain viruses, heavy metal ions, large molecules and very small particles. The process is used for water softening and in drinking water treatment.
The Ultrafiltration is an important process for drinking water and wastewater treatment. It is used to separate particles, microorganisms, proteins and turbidity from the water, including in the membrane bioreactor (MBR). Ultrafiltration is used, for example, to purify circulating water in swimming pools. As the formation of clogging deposits on the membrane can now be avoided, existing wastewater treatment plants are increasingly being supplemented with ultrafiltration as a polishing step. When retrofitting older, conventional wastewater treatment plants, ultrafiltration can be used directly in or after the aeration tank to replace downstream treatment steps or to increase the purification performance of biological wastewater treatment.
The Reverse osmosis is an important process step, for example in the concentration of landfill wastewater, in drinking water treatment in rural regions that are not connected to the mains, in seawater desalination or in the decalcification of boiler water in power plants. The concentration of substances dissolved in liquids is increased through a semi-permeable membrane by using pressure to reverse the osmosis process: If the pressure is higher than the respective osmotic pressure, the molecules of the solvent diffuse to the side of the membrane on which dissolved substances are already less concentrated. This process is also used to obtain ultrapure water.
Other Important Chemical-Physical Methods

Wastewater Treatment by Flotation
In flotation, dispersed or suspended substances are transported from liquids to the surface with the help of small gas bubbles and removed there using a scraper device. Flotation processes are used in wastewater treatment to separate oils, fats and finely suspended solids, such as suspended solids.
The smaller the microbubbles are, the better the particles or droplets accumulate. For this reason, dissolved air flotation (DAF) is often used in wastewater technology. It has proven itself due to its cost-effectiveness and efficiency. Flotation processes can also be supported by the use of auxiliary materials such as collectors, frothers, regulators or pushers.
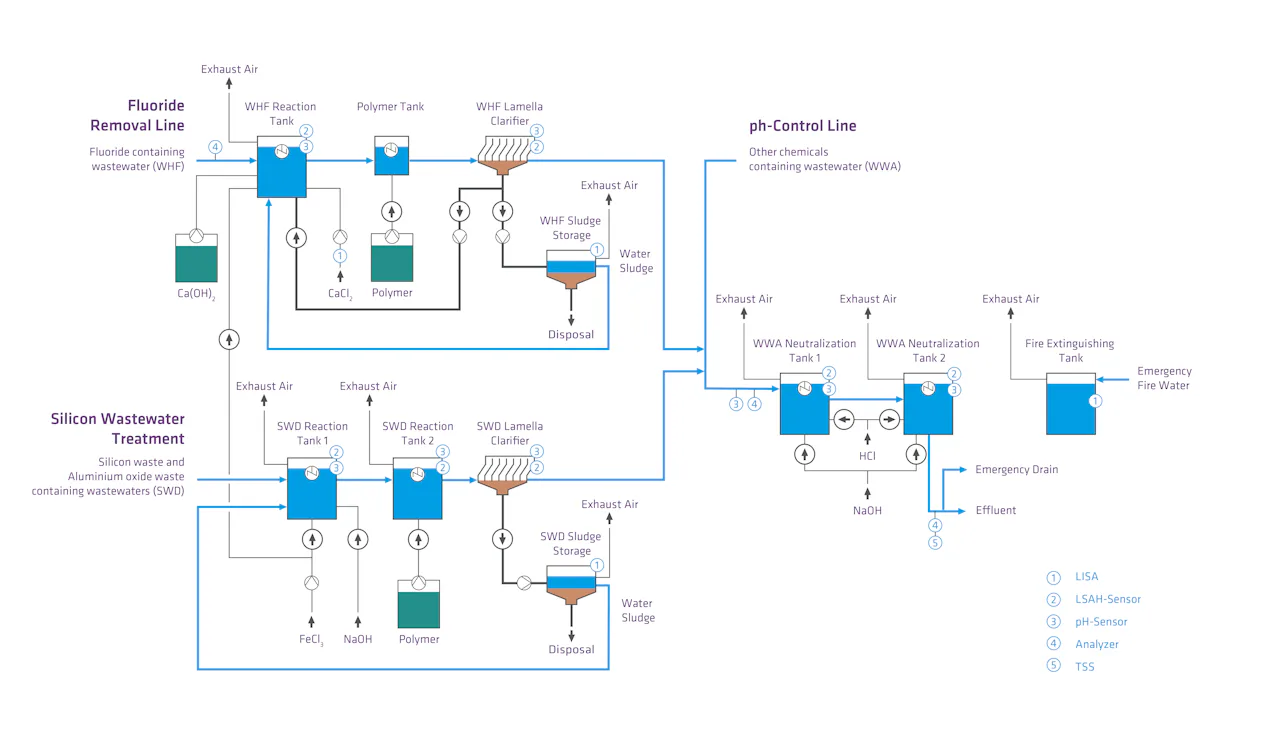
Project Report: Innovative Chemical-Physical Water Treatment for the Semiconductor Industry
DAS Environmental Experts has implemented a state-of-the-art wastewater treatment system for a customer in the semiconductor industry that meets the highest standards of process reliability, purity and resource efficiency. Semiconductor production places particularly high demands on wastewater technology, as even the slightest impurities can impair production processes.
Detailed advance planning was a key success factor in the project. It enabled the precise design of the plant, the reliable removal of all relevant pollutants, the optimization of the use of resources, compliance with international environmental standards and rapid adaptation to local conditions.
The system combines state-of-the-art technologies in three parallel lines:
Fluoride treatment to safely reduce the fluoride concentration
CMP wastewater treatment for the removal of silicon, aluminum and other solid particles
Neutralisation for reliable adjustment of the pH value
This project shows how modern chemical-physical processes can be used effectively in the semiconductor industry to meet the highest environmental, safety and quality standards.
The system was designed for high operational reliability, efficient processes and maximum productivity:
Continuous operation for maximum availability
Fully automated system operation with minimal personnel deployment
Compact design for optimum use of the available space
Guaranteed discharge values
The wastewater system ensures stable and legally compliant discharge conditions:
Fluoride ≤ 10 mg/L
TSS (Total Suspended Solids) ≤ 300 mg/L
pH value 6-11
Combination of State-of-the-Art Technologies for the Effective Chemical-Physical Treatment of Process Wastewater
Looking for support in choosing the right chemical-physical process?
We can help you select and optimize the right treatment procedures. Contact our team of experts.
Deputy Chief Operating Officer Water Treatment